

Jonna van der Stam
Projects: Heart Failure Classification Trial (HaFaC) & Transitional Care Trial (TRICA)
More and more sources of clinical information, e.g. novel biomarkers and wearable sensors, are becoming available. To make the transition from data to application in clinical practice, my projects focus on potential novel markers throughout the hospital, ranging from the outpatient clinic to the general ward and intensive care unit. In the outpatient clinic, the Heart Failure Classification (HaFaC) Trial explores objective laboratory biomarker-based classification with the aim to aid cardiologists in differentiating high risk from low risk heart failure patients. In the general ward and intensive care unit, the Transitional Care (TRICA) trial explores the potential of a novel wearable sensor HealthDot (Philips). The project focusses on accuracy of the measured vital parameters in real world patients, as well as their potential for deterioration detection in postoperative patients. As a technical PhD with a background in Medical Engineering based in the Clinical Laboratory of the Catharina Hospital and TU/e, I work closely together with PhD’s with a Medicine background as well as hospital specialists and industry partner Philips Research. Within these collaborations we combine different areas of expertise which allows us an work on clinically relevant research, aiming to improve care for future patients.
Juliette van Haren
Project: Designing for Perinatal Life Support technologies through simulation
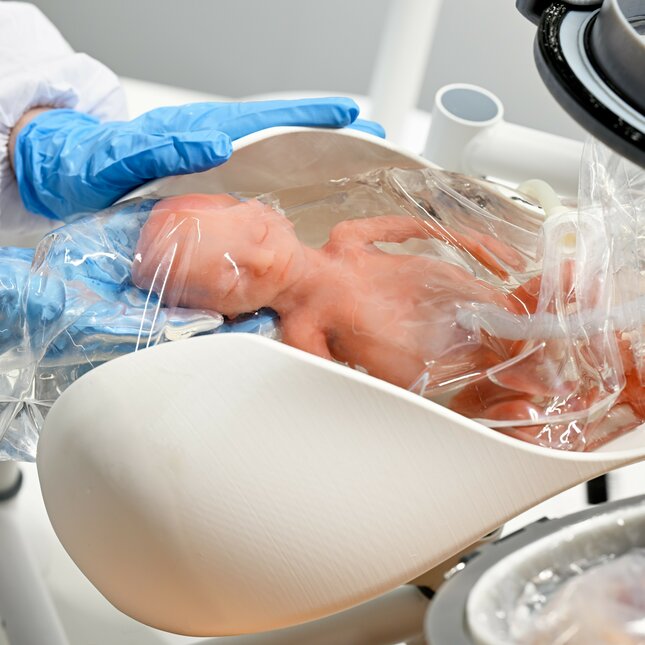
Every year, 15 million children are born prematurely worldwide, of which 5% at extremely premature age (born before 28 weeks of gestation). Current treatments in neonatal intensive care units (NICUs) are suboptimal for the fragile and immature organs of these neonates. The Perinatal Life Support (PLS) project aims to develop an artificial womb (PLS system) that retains the intra-uterine circumstances extracorporeally, i.e. a liquid-based environment to facilitate fetal development ex utero. Our research focuses on the use of simulation technologies to develop and validate PLS technologies. One aspect that we are developing is the transfer procedure, which aims to provide a smooth transfer from the native uterus to the PLS system and prevents the infant from breathing (see figure). As a result of the development of a smart fetal manikin, with embedded sensors and actuators, we can perform user studies and medical training through interactive simulations. This fetal manikin is in contact with a digital twin, developed by e/MTIC PhD Bettine van Willigen at the dept. of Biomedical Engineering, and allows for a data-driven approach to design. In a simulated operating room all prototypes and manikins come together, and we can co-create and validate with medical specialists from Máxima Medical Center.
David Melo
Project: UMOSA: Laser-based illumination unit for rPPG
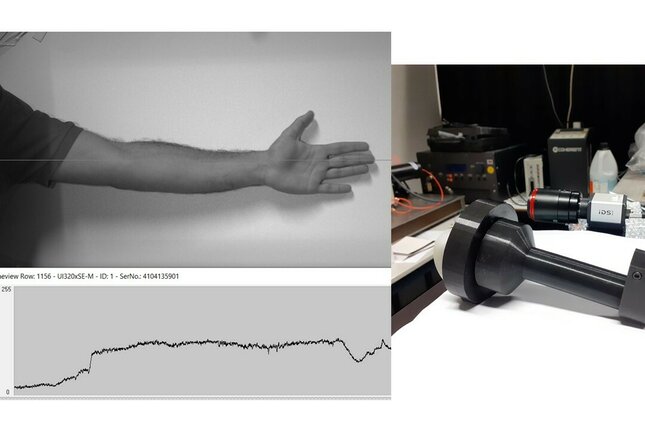
In the past few years, Remote Photoplethysmography (rPPG) was proven a valid solution for contactless monitoring of vital signs. The UMOSA project aims at investigating how this technology can improve the current sleep apnea diagnosis methods. Within the UMOSA, a laser-based, modulated, stable, and efficient light source was built to expand the number of vital signs that are possible to monitor with rPPG and/or the number of cameras required to collect the signal. After concluding the implementation, the light source is ready to be tested and the improvements it might bring will be thoroughly investigated at TUe during the next months. The UMOSA consortium is formed by TUe, Philips Research, and the Kempenhaeghe Sleep Center. Kempenhaeghe, on the clinical side, provides knowledge regarding how clinical staff and patients would look at the prototype while Philips brings technical knowledge and helps in the understanding of how the industry evolves. Finally, TUe is responsible for providing support and technical guidance. As a PDEng trainee, the 3 entities were extremely important so I could grasp the entire picture.Under the UMOSA proje ct, 2 other contactless technologies for the diagnosis of sleep apnea (Speckle Vibrometry and Remote Thermography) are also being investigated.
Chenyan Huang
Project: PICASSO – Maternity Dashboard
The dashboard is widely used in various areas to provide a visual display of key performance information for better operational decision-making by summarizing and integrating related data across an organization. Similarly, a maternity dashboard is expected to provide the clinicians with the essential information in time to assist the daily decisions and improve healthcare quality in the maternity unit. Currently, most maternity dashboards were created based on the medical knowledge and experiences of the medical professionals instead of the EMR data in the dataset system. To massively improve the accuracy and effectiveness of the maternity dashboard, this research will use the EMR/EHR data from clinical systems to obtain novel measurements and bench markers for the maternity dashboard. In this study, data-driven approaches can be expected to aggerate data from multiple sources, implement reliable data infrastructure, and for further analytics, the proper indicators will be selected and the threshold values will be calculated by data mining, and finally, the information from EHR/EMR data will be intuitively displayed/visualized on one dashboard and enable clinical excellence to flourish. This research is supported by e/MTIC partners TU Eindhoven, Maxima MC, and Philips Research, which enables a comprehensive collaboration of expertise from multiple disciplines.

Cindy Verstappen
Project: FORSEE project: video monitoring FOR early Signaling of adverse EvEnts
Generally, clinical staff on the hospital ward collect vital signs at regular intervals to determine a patient’s cardiorespiratory status. This spot-check approach has clear limitations. First, the interval between spot-checks is undesirably long, as deviations in vital signs that could indicate acute adverse events typically take place on a shorter timescale. Furthermore, the approach depends on contact sensors, such as ECG leads, which can be uncomfortable for the patient, especially during sleep. As part of an User Centered Design (USD) in the FORSEE project, we conducted semi structured questionnaires on the topics privacy, wireless monitoring and requirements of the system among professionals (users) on general wards of cardiology, cardio thoracic surgery, surgery and Emergency Cardiac Care (ECC). Moreover, patients at ECC undergoing cardioversion were interviewed about their experiences with topics such as privacy and level of comfort with this VBM. We use an UCD to arrive at a usable system; relevant and meets the wishes of the patient and health care provider. The continuous collaboration between clinical PhD, technical PhD, clinical nurse practitioner and all other stakeholders works efficiently. In our project we work also with other (PhD)students and students of Fontys University of Applied Sciences. The FORSEE project is a collaboration between Eindhoven University of Technology, Phillips, e/MTIC, Fontys University of applied Sciences and the Catharina Hospital Eindhoven.

Steven Beumer
Project: PerStim
Approximately 30 % of all epilepsy patients do not respond to anti-epileptic medication. This type of epilepsy is called refractory epilepsy. For some of these patients, brain surgery is usually the only conventional treatment option that remains. This is highly invasive, risky, and is not feasible for all patients due to the complexity of the disease. In the PerStim (Personalized Neurostimulation) project we would like to explore transcranial neurostimulation as a treatment for this group. In this e/MTIC consortium we work together with Kempenhaeghe, Philips and the university hospital of Ghent to optimally combine clinical and technological knowledge. The e/MTIC ecosystem allows us to quickly get feedback on clinical protocols and data transfer agreements. At the university we work on electrical field modelling and personalization of the stimulation combined with phantom experiments to validate our simulations. In the medical centers, clinical trials are done on healthy volunteers as well as patients. In these trials the technical and clinical knowledge will be combined to both investigate the efficacy of the stimulation and perform pilot studies on the intended patient population. Infographic made by Eindhoven Engine

JAAP VAN DER AAR
PROJECT: COMBAT-VT: PROLONGED IN-HOME SLEEP MONITORING (PRISM)
Since the 1960s, polysomnography (PSG) has been considered as the gold standard for sleep diagnostics. PSG enables to simultaneously measure a wide variety of physiological signals related to sleep, including eye, brain, muscle, and cardiac activity, but also requires a minimum of 22 wires attached to the patient. In addition, PSG is often performed over one night in an artificial clinical setting, which negatively affects one’s sleeping experience and is unable to expose night-to-night variability.
We recently started the prolonged in-home sleep monitoring (PRISM) study, which aims to investigate how we can unobtrusively measure sleep in the disordered population. For seven nights, the patients’ sleep is monitored using a watch with photoplethysmography (PPG) and actigraphy, bed pressure sensors, a wearable electroencephalography (EEG) headband, and a digital sleep diary. We aim gather more information on how sleep in patients varies, which modalities are necessary to capture (abnormal) sleep, and how home monitoring can be beneficial as additional instrument.
This study shows the strength of e/MTIC and what can be achieved with cooperation. Only with the access to patients through Kempenhaeghe, the research know-how of the TU Eindhoven, and the use products from Philips, such a study is possible.
Carlijn Buck
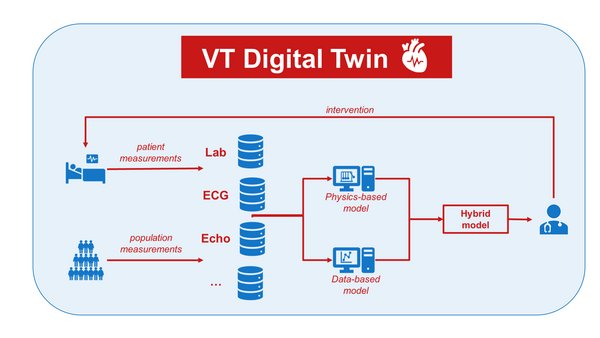
Project: COMBAT-VT: Digital Twin to Predict Ventricular Tachycardias using Hybrid Modelling
Post-myocardial infarct patients have an increased risk for scar-based ventricular tachycardia (VT), an abnormal heart rhythm, which may lead to sudden cardiac death (SCD). An implanted cardioverter defibrillator as well as ablation can prevent SCD. However, the current indications for both treatments proves to be poor in discriminating between patients with a high or low risk of SCD.
COMBAT-VT aims to improve the understanding and prediction of VTs and provide optimized patient-specific therapy guidance. My own project focusses on developing a VT Digital Twin with data and medical knowledge of the Catharina Hospital and modelling knowledge of Philips and TU/e.
In this project we take a hybrid modelling approach. As a first step, Physics Informed Neural Networks (PINNs) are used. Here the loss of the neural network (data-driven model) with time as input and left ventricular pressure as output, is depending on two matches: 1) predicted pressure with the labeled pressure; 2) predicted pressure with the one-fiber model equation (physics-based model). This ensures the neural network is regulated with known physics. The first preliminary results show that with small data sets training is more efficient and yields better results if physics is included in the network.
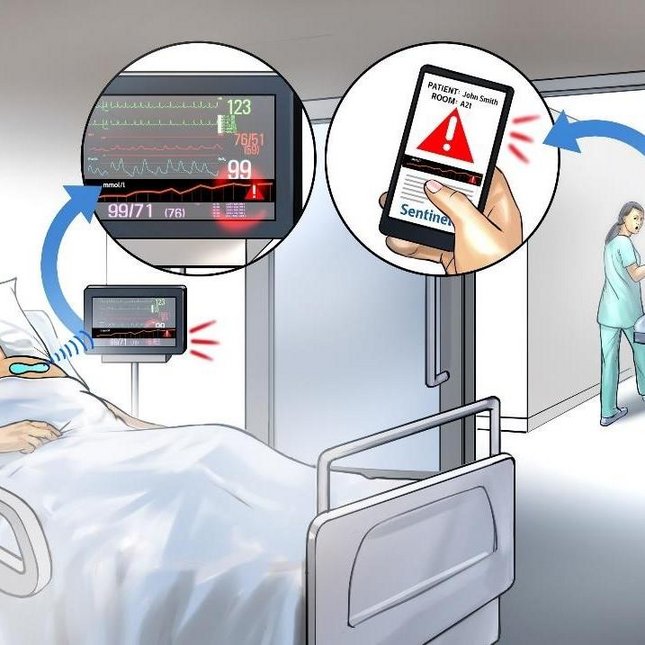
Emma Moonen
Project: Sentinel - biomarker monitoring by an autonomous wearable patch
Every year thousands of patients in Europe and elsewhere die or suffer serious complications and/or disabilities following ‘adverse events’ during their stay in hospital or after discharge. The Sentinel project aims to reduce these risks to patients through an improved wearable early warning system (EWS) – a ‘patch’ – that detects when a patient’s condition is deteriorating. Adverse events are estimated to affect up to 12% of hospitalized patients and to cause up to 95,000 patient deaths per year in the European Union alone. Current EWSs measure physiological vital signs (such as blood pressure and respiration rates) and levels of consciousness. However, clinicians consider that biomarkers contained in sweat can also be valuable indicators of deteriorating condition. Efforts have been made to include these markers to improve existing EWSs, either through point-of-care (POC) testing or increased central lab testing. However, the lack of integration into caregiver workflows has prevented widespread take-up. Sentinel is addressing these issues by realizing significant innovations in sweat rate and biomarker sensing technologies, that will enable a wearable solution based on semi-continuous quantitative sensing of all three indicators: physiological, contextual and biomarker. The project will focus on two key use cases, sepsis and delirium. And amongst a range of innovations, it will work on advances in micro/nano technologies and microfluidics, as well as manufacturing technologies and verified prototypes. Noteworthy is the research towards utilizing sweat as biomarker-rich biofluid with fully non-obtrusive access, however the sweat rate especially in sedentary state is very small, in the order of 0.2 nL/min per gland, insufficient to transport sweat even in small microfluidic channels in a clinically relevant timely manner to the biosensors. Sentinel is approaching this dilemma by an innovative active transport mechanism that can even accelerate the smallest amounts of sweat. e/MTIC partners TU/e, Philips and the Catharina Hospital are part of the Sentinel consortium. Within the e/MTIC ecosystem we can get quick feedback from the hospital to optimize the design and accelerate the development.

IRIS CRAMER AND RIK VAN ESCH
PROJECT: FORSEE-PROJECT (VIDEO MONITORING FOR EARLY SIGNALING OF ADVERSE EVENTS)
The FORSEE project explores continuous video monitoring of the cardiorespiratory status of a patient as an innovative unobtrusive method that could aid to reduce workload for the clinical staff, improve patient comfort and better predict deterioration to allow early intervention and potentially save lives.
The first clinical study investigated the performance of the video monitoring technology (based on the principle of remote photoplethysmography) to detect heart rate and respiration rate in a population with cardiac arrhythmias. First results show that video monitoring provides accurate measurements of heart rate during sinus rhythm, atrial fibrillation and atrial flutter. This motivates towards a development of an automated algorithm to distinguish between these rhythms, enabling early cardiac arrhythmia detection.
In the FORSEE-project, a medical PhD and a technical PhD collaborate on a daily basis to bring the video monitoring technology from healthy volunteers in a lab setting to real patients in the clinic. They are supervised by a multidisciplinary team of researchers of TU/e, Catharina hospital, Fontys and Philips Research, which brings a wide range of expertise together. Moreover, research on patient- and user experiences is integrated within FORSEE.

MELISSA NIEMANTSVERDRIET
PROJECT: DESIGN OF A CLINICAL DATA MANAGEMENT WORKFLOW FOR THE COMBAT-VT PROJECT
The clinical data management workflow is a standardized process to support large-scale medical (data-driven) research. It is developed in the Catharina Hospital and is applied in the COMBAT-VT [link: www.combatvt.nl] project. In this project, multiple PhD researchers Qualified Medical Engineer, PDEng researcher at the TU/e are developing models that aim to predict the risk of ventricular tachycardia (heart rhythm disorder) accurately following a myocardial infarction. Furthermore, outcomes of patient-specific treatment strategies will be predicted using a Digital Twin concept. The models require large amounts of input data in order to optimally train and validate the models. These data originate from multiple sources within the hospital. In the workflow, the data are obtained in bulk and made accessible according to the FAIR principles. More importantly, the data must not be traceable to the patient when made accessible to the researchers within the project, but still contain sufficient information to perform patient specific predictions. In a pilot, structured datasets were created consisting of available lab results, vital parameters, echocardiograms, and ECGs of 46 patients. This project was part of the e/MTIC collaboration, which enabled the possibility to share data with the PhD researchers in anDREa, a protected digital research environment. They are currently testing their models with the data and the clinical data management workflow.