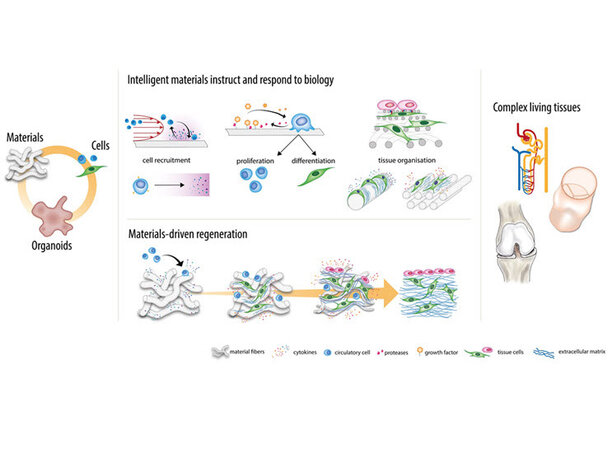
Modelling and experimentation at length scales from molecule to man
While performing our research at the forefront of soft tissue biomechanics, mechanobiology, and the engineering of living soft biological tissues and organs, we strive to provide a stimulating educational environment for graduate and post-graduate students. Our research is multi-disciplinary at heart, combining concepts from molecular, cell and tissue biology, (patho)physiology, immunology, biomechanics, physics, engineering, and materials science. To translate our fundamental knowledge to (bio)medical applications we actively collaborate with clinical partners, industry, and patient organizations.
Read moreMeet some of our Researchers
Research Lines
News

March 15, 2024
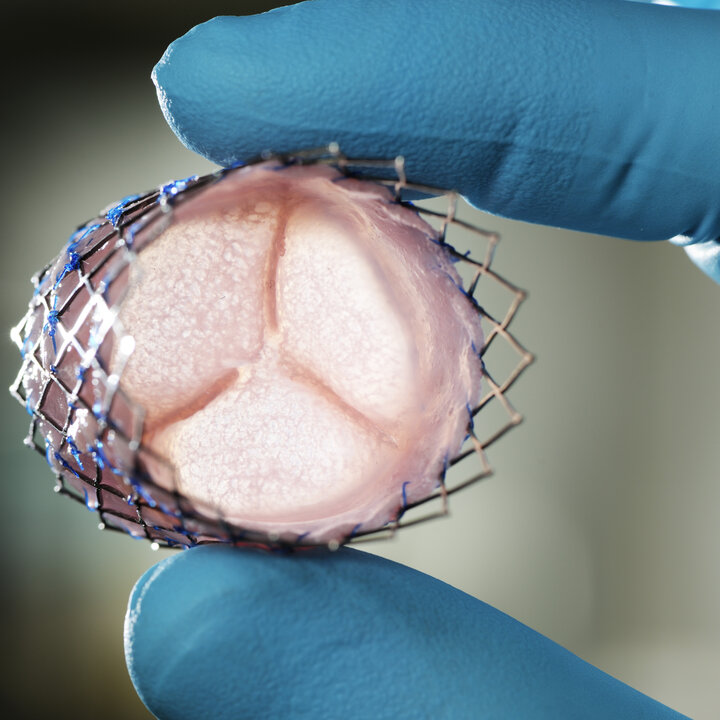
Blood-friendly inner lining for artificial hearts
Dina Ibrahim defended her thesis at the Department of Biomedical Engineering on March 5.
Read more
![[Translate to English:] [Translate to English:]](https://assets.w3.tue.nl/w/fileadmin/_processed_/7/e/csm_Bril%20Banner%20image_0d3235e44f.jpg)
September 29, 2023
Shining a new light on cell response in the body
Researchers develop a new, light-responsive hydrogel upon which cells can grow to study how cells deal with changes in their environment.
Read more
Recent Publications
Our most recent peer reviewed publications
-
Joshua A. Welsh,Deborah C.I. Goberdhan,Lorraine O’Driscoll,Edit I. Buzas,Cherie Blenkiron,Benedetta Bussolati,Houjian Cai,Dolores Di Vizio,Tom A.P. Driedonks,Uta Erdbrügger
Minimal information for studies of extracellular vesicles (MISEV2023)
Journal of Extracellular Vesicles (2024) -
Elana M. Meijer,Rachel Giles,Christian G.M. van Dijk,Ranganath Maringanti,Tamar B. Wissing,Ymke Appels,Ihsan Chrifi,Marianne C. Verhaar,Anthal I.P.M. Smits,Caroline Cheng
Effect of Mechanical Stimuli on the Phenotypic Plasticity of Induced Pluripotent Stem-Cell-Derived Vascular Smooth Muscle Cells in a 3D Hydrogel
ACS Applied Bio Materials (2023) -
Freddy Suarez Rodriguez,Sami Sanlidag,Cecilia Sahlgren
Mechanical regulation of the Notch signaling pathway
Current Opinion in Cell Biology (2023) -
Janine Grolleman,Nicole C.A. van Engeland,Minahil Raza,Sepinoud Azimi,Vito Conte,Cecilia M. Sahlgren,Carlijn V.C. Bouten
Environmental stiffness restores mechanical homeostasis in vimentin-depleted cells
Scientific Reports (2023) -
Annemijn Vis,Bente J. de Kort,Jan Willem van Rijswijk,Rob Driessen,Niels Wijkstra,Paul F. Gründeman,Hans W.M. Niessen,Henk M. Janssen,Serge H.M. Söntjens,Patricia Y.W. Dankers
Evaluation of pliable bioresorbable, elastomeric aortic valve prostheses in sheep during 12 months post implantation
Communications biology (2023)
PhD Research Projects
Contact
-
Postal address
P.O. Box 513Department of Biomedical Engineering Eindhoven University of Technology5600 MB EindhovenNetherlands -
Visiting address
Building 15, Gemini-South (room 4.115) Groene LoperEindhoven University of Technology5612 AZ EindhovenNetherlands -
Secretary
![[Translate to English:] Frank Baaijens at ESB 2023. Photo: Sandra Loerakker](https://assets.w3.tue.nl/w/fileadmin/_processed_/8/6/csm_Baaijens%20ESB2_6679d2e011.jpeg)
![[Translate to English:] Bente de Kort](https://assets.w3.tue.nl/w/fileadmin/_processed_/5/8/csm_de_Kort_Bente_BMT_VH_9192_PhD_7c6894c5e6.jpg)